How much time and effort does your team spend identifying and comparing similar clinical trials to optimize study design?
Are you confident that your clinical trial eligibility criteria are maximizing patient recruitment without unnecessary exclusions?
How do you currently integrate real-world evidence into trial design to ensure alignment with actual patient populations?
What challenges do you face when justifying trial designs to regulatory agencies based on historical evidence?
How do you currently track competitor clinical trial strategies, and how often do you reassess your trial designs based on industry trends?
Answering these questions highlights how vital clinical trials are in a drug developmental cycle. Trial design and start up activities can be improved and accelerated with the use of AI to automate the identification and qualification of trial candidates, and the analysis of competitor trials. Research teams must be able to distill the most accurate, up-to-date information by disambiguating across mountains of content from clinical registries, scientific publications, and real-world data.
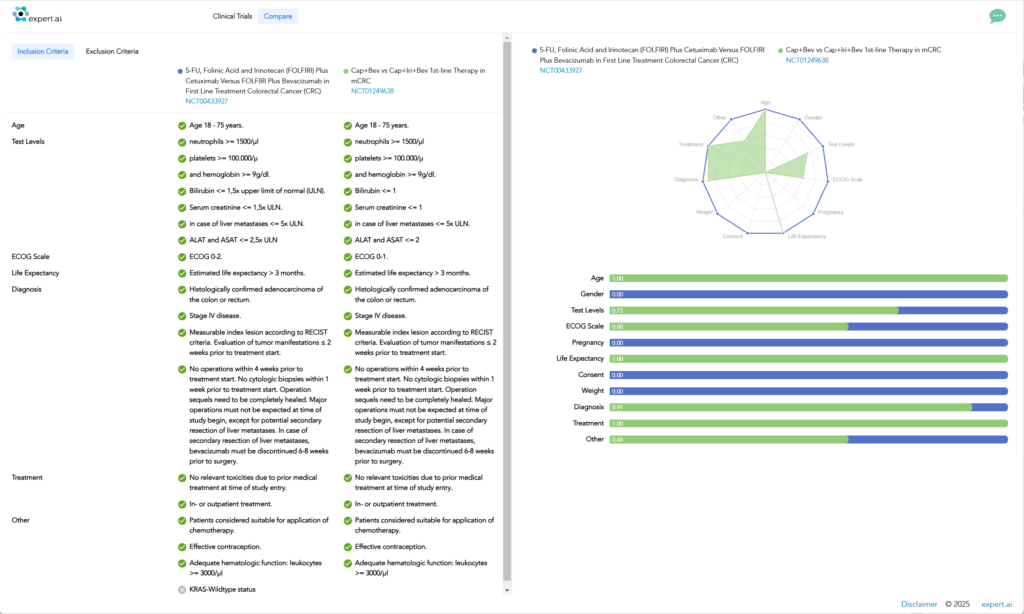
One of the most important criteria for clinical trial design and success is related to patient recruitment. Expert.ai’s hybrid AI capabilities make it possible to navigate through millions of records simultaneously, extract real-world data from unstructured data sources, create key attributes for a patient profile, and identify site locations. Allowing trial protocol requirements to be matched against real-world evidence provides insights to optimize eligibility criteria, reduce timeline risk and speed up patient enrollment.
The imperative for rapid innovation in pharma fuels evolving treatments to keep the pipeline rich and drives fierce competition in the industry. This in turn requires keeping a close watch on the competition with a robust pharma competitive intelligence strategy that includes the monitoring of clinical trials and scientific literature sources for insights into ongoing drug developments.
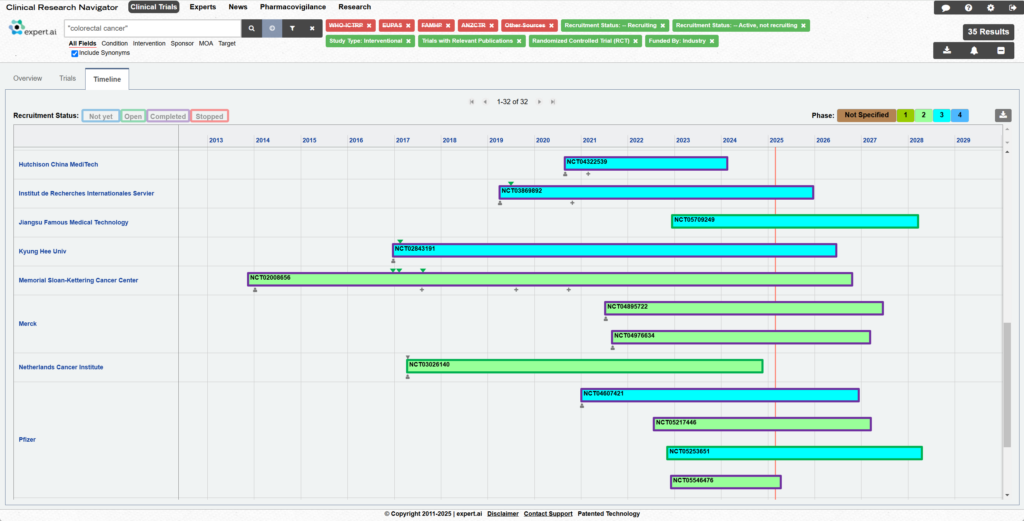
Expert.ai’s technology mines data from more than 900,000+ clinical trials worldwide. This includes clinical trial registries such as clinicaltrials.gov, EUDRA, EUPAS, Japanese registries, Australian registries, and others. It provides the most up-to-date and comprehensive data landscape by disease, drug, mechanisms of action, organization, or geography. Not only can researchers find related trials, but they can also access related publications, news, study results and principal investigators all in one place.
Data Indexing & Classification
Utilize AI to structure and classify publicly available data on a targeted disease area.
Organize and filter scientific publications and clinical trial records to extract relevant insights.
Eligibility Criteria Matching
Identify and classify trials based on specific inclusion/exclusion criteria.
Enable efficient mapping of patients or trial designs to relevant research.
AI-Powered Information Retrieval & Reasoning
Implement generative AI to answer research questions on the fly.
Bridge structured (tables, clinical trials data) and unstructured (scientific literature) information sources.
Agentic AI for Continuous Learning & Insights
Continuously monitor and integrate new scientific data.
Highlight changes in insights over time, mimicking self-reasoning and adaptive learning.